Looking to the Retina
The motor center of the brain (Substantia Nigra) is made up of dopamine producing nerve cells. These cells are degenerated in Parkinson’s disease (PD). This degeneration underlies most of the motor features of PD and has been a therapeutic focus since the 1960s. Levodopa enhances dopaminergic neurotransmission along the neuronal pathway from the substantia nigra (nigrostriatal pathway) and relieves many of the motor features of PD.
Even with Levodopa and best medical care, most non-motor problems persist, and motor symptoms continue to worsen over time in PD.
Related deterioration in the Retina
Neurodegeneration in PD also affects the eye and leads to major health consequences.
In front of the rods and cones lie the retinal ganglion cells which contain a photoreceptor called melanopsin. These melanopsin containing retinal ganglion cells (mRGCs), are different than rods and cones, and they send non-visual signals to various areas of the brain involved with sleep, alertness, mood, and cognition.
Light is absorbed by the mRGCs and signals through a neuronal path (the retinohypothalamic tract) to regulate circadian rhythms. Normal circadian function is critical to human health because it regulates nearly all of our physiological function.
Recent scientific work has established that mRGCs are deteriorated in PD and in other neurodegenerative diseases. As a result, the normal signaling pathway is deficient. Researchers have observed associations between circadian disruption and symptoms such as sleep disturbances, fatigue, mood changes, and cognitive challenges in people living with Parkinson’s disease.
Health, Circadian Rhythmicity and PD
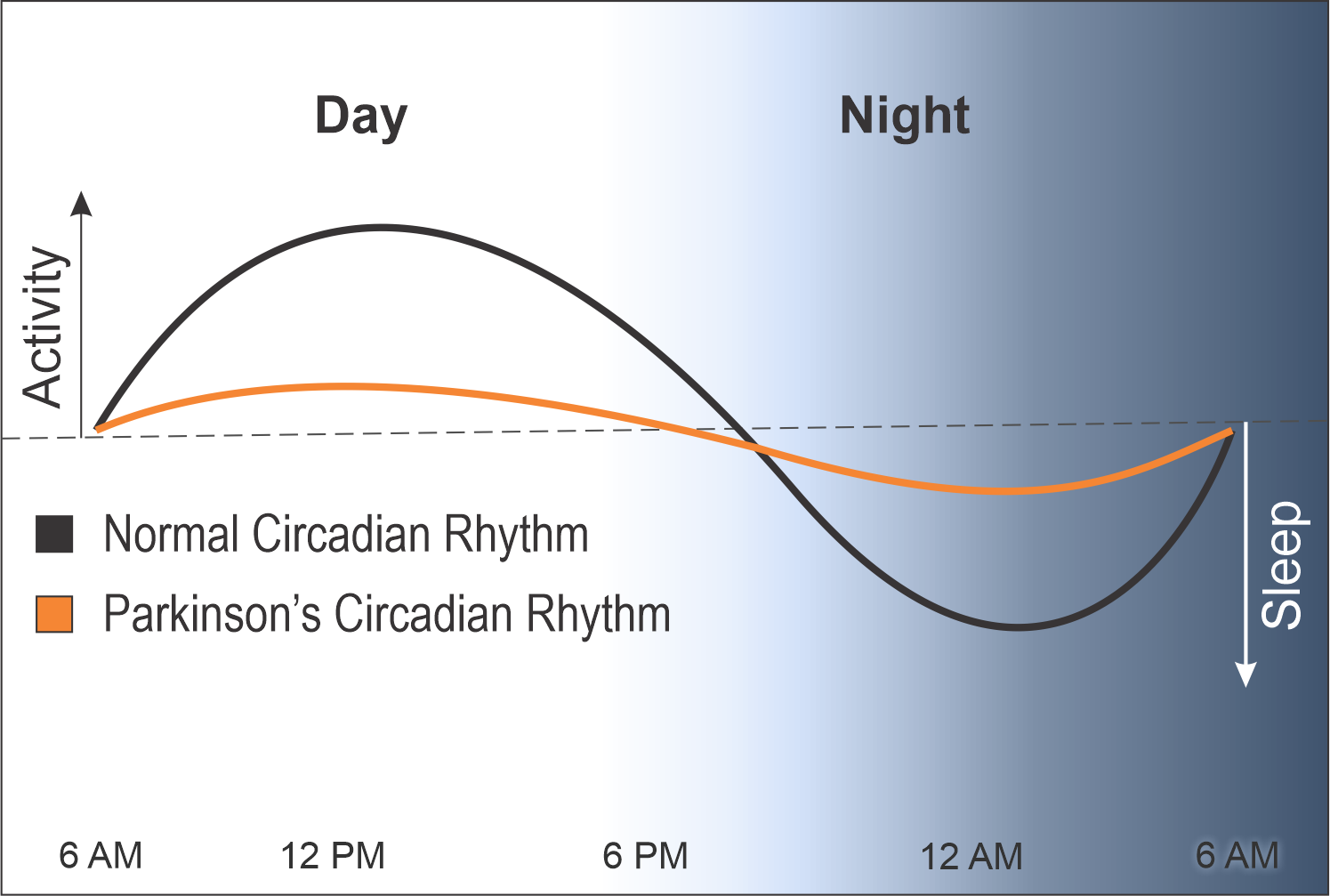
A normal circadian rhythm is measured by hormonal release and gene expression. The peaks in the day represent energy and activity and the troughs at night represent healthy sleep and rest.
In contrast, the circadian rhythms of people with PD are heavily blunted, causing them to have less energy during the day and poor sleep at night.
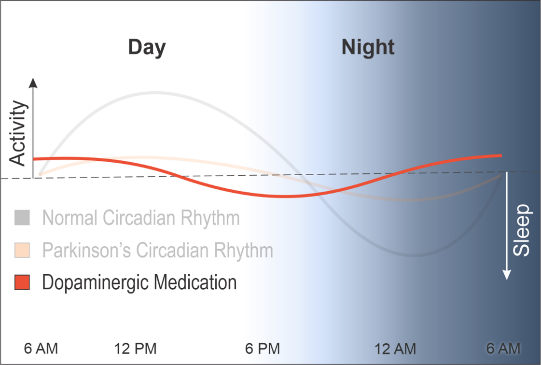
To make matters worse, the dopamine medications used in PD push this blunted rhythm earlier, causing people with PD to get tired earlier in the day and to wake at night, further disrupting their rhythms and exacerbating physical and cognitive decline.
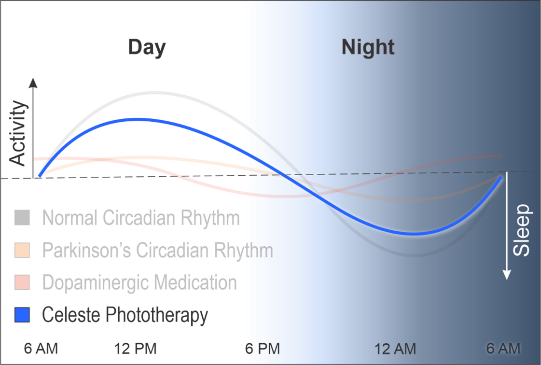
Researchers have investigated whether targeted light exposure may influence melanopsin signaling and circadian function. The potential clinical implications of these biological effects continue to be evaluated through ongoing scientific research.
Celeste Photo-Neuromodulation
Research has shown that intrinsically photosensitive retinal ganglion cells (mRGCs) play an important role in transmitting light-responsive signals to areas of the brain involved in circadian regulation and neurological function. Studies have also suggested that these pathways may be altered in Parkinson’s disease.
PhotoPharmics has developed Celeste, an investigational photo-neuromodulation device designed to explore whether specific wavelengths, intensities, and timing of light can influence these biological pathways. The device incorporates proprietary light delivery technology developed from research into retinal signaling and circadian biology.
Celeste delivers light passively through the eyes during a seated session and is designed for home use under physician supervision as part of clinical research and evaluation. Clinical studies have been conducted to better understand the relationship between photo-neuromodulation, retinal signaling, circadian biology, and Parkinson’s disease.

Supporting Articles
Circadian rhythms are the daily activity and sleep patterns regulated by the suprachiasmatic nucleus, and they have a profound effect on daily physiological function. These rhythms are reset daily in response to light.
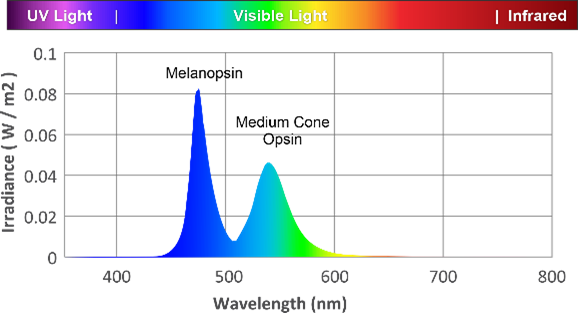
Melanopsin is the primary signaler for circadian regulation. Melanopsin containing retinal ganglion cells (mRGCs) are required for non-image signaling, such as circadian regulation. Rods and cones can contribute to non-visual signaling, but this process is governed by the mRGCs.
- Berson DM, Dunn FA, Takao M. Phototransduction by retinal ganglion cells that set the circadian clock. Science 2002;295:1070-1073. Güler, A. D., et al. (2008).
- Melanopsin cells are the principal conduits for rod–cone input to non-image-forming vision. Nature, 453(7191), 102–105.
Circadian system responds to specific wavelengths of light. Light at low intensities stimulate rods and cones, which activate melanopsin signaling, however sustained non-visual signaling occurs via 480nm excitement of the melanopsin photoreceptors, at higher irradiance than rods and cones.
Circadian rhythms are blunted in Parkinson’s and do not respond to normal light signals. Melanopsin cells are damaged in neurodegenerative diseases, resulting in blunted circadian profiles.
- La Morgia, C., et al. (2017). Retinal Ganglion Cells and Circadian Rhythms in Alzheimer’s Disease, Parkinson’s Disease, and Beyond. Frontiers in Neurology, 8, 162. Lax, P., Ortuño-Lizarán, I., Maneu, V., Vidal-Sanz, M., & Cuenca, N. (2019).
- Photosensitive melanopsin-containing retinal ganglion cells in health and disease: Implications for circadian rhythms. International journal of molecular sciences, 20(13), 3164.
- Evans JA, Davidson AJ. Health consequences of circadian disruption in humans and animal models. Prog Mol Biol Transl Sci. 2013;119:283-323. doi: 10.1016/B978-0-12-396971-2.00010-5. PMID: 23899601.
Additional evidence of damaged melanopsin signaling. Melanopsin signaling also controls the sustained pupillary light reflex which is damaged in neurodegeneration.
- Joyce, D. S., et al. (2018). Melanopsin-mediated pupil function is impaired in Parkinson’s disease. Scientific Reports, 8(1), 1–9.
- Chougule, P. S., Najjar, R. P., Finkelstein, M. T., Kandiah, N., & Milea, D. (2019). Light-induced pupillary responses in Alzheimer’s disease. Frontiers in neurology, 10, 360.
Scientific Context
The biological mechanisms described on this page form the basis of ongoing research. Celeste® is an investigational device, and clinical benefit has not been established.
